This article is featured in the 2021 Commencement special issue.
Since the start of the pandemic, the creation and distribution of safe and effective vaccines has dominated public discourse. As of Tuesday, 42% of Americans and 57% of Dartmouth community members have been fully vaccinated against COVID-19, and the ongoing vaccination campaign reflects the critical role vaccines play in combating infectious diseases. The College’s own history with vaccines is storied, and ongoing research at Dartmouth continues to lead to the creation and testing of groundbreaking vaccines.
A history of vaccines
“When we look at the history of public health, particularly in the twentieth century, safe, effective and scalable vaccines are one of the most important accomplishments of the global public health community,” government professor and vaccine policy expert Herschel Nachlis said.
So how do vaccines actually work? When a pathogen — a bacterium, virus, parasite or fungus — enters the body, the immune system develops specific antibodies in response to the antigens contained on the pathogen. Before these antibodies are developed, a person can fall ill or die from the disease caused by the pathogen. If a person is infected with the same pathogen later, their body can produce the necessary antibodies more rapidly and prevent the disease from overtaking the body’s defenses.
Vaccines cause the body to develop these antibodies without infecting the recipient of the vaccine with the disease. For example, the Pfizer-BioNTech and Moderna vaccines use messenger RNA to teach human cells how to build proteins that trigger an immune response; the Johnson & Johnson vaccine, in a more traditional method, uses an inactivated adenovirus to convey these instructions to cells.
While vaccines as they are known today are a modern development, the practice of inoculation dates back centuries and has historically focused primarily on smallpox, the now-eradicated infectious disease responsible for killing 300 million people in the twentieth century alone. An early method for preventing smallpox — and a precursor to vaccination — was variolation, a process by which people who had never had the disease inhaled or scratched smallpox sores into their arm with the hope of contracting only a weak case of smallpox and developing immunity as a result. It was not until 1796, when British physician Edward Jenner found that the mild cowpox virus granted immunity to the much more deadly smallpox, that the groundwork for vaccine development was laid. After his discovery, Jenner coined the word “vaccine” — a derivative of the Latin words for cow, “vacca,” and cowpox, “vaccinia.”
Vaccines at Dartmouth
Despite smallpox’s constant threat at the time, College founder and former president Eleazar Wheelock, an asthmatic who was especially fearful of infection, in 1776 refused to inoculate Dartmouth students. However, local physician Laban Gates did so anyway. Wheelock saw inoculation as “communicating the small pox” among his community due to the increased danger of providing more hosts for the virus. In response to Wheelock’s push against inoculation, students and trustees drafted a petition accusing him of “injuriously encroach[ing] upon the privileges and immunities of this College.”
More recently, Dartmouth has seen outbreaks of the potentially fatal meningitis, the illness caused by meningococcus bacteria. Throughout 1995, several students contracted meningitis, and at that year’s commencement, a student exposed former U.S. President Bill Clinton, that year’s commencement speaker, to the illness.
In June of 1999, a junior died of meningitis at the end of spring term, and another was infected but recovered, causing fear of a possible outbreak. At this time, the College only recommended — but did not require — meningitis vaccines for students, who are at increased risk of contracting the disease due to the community setting of dormitories. The College announced that any students scheduled to live in-residence during the summer term could request to have the summer residency requirement waived, and even considered canceling the entire summer term, but the state health department advised that such a measure was unnecessary.
In response to this outbreak and advice from the New Hampshire State Health Department, the College provided free meningococcal vaccines to students. The program eventually vaccinated more than 1400 students, or 80% of students enrolled for the summer term. The vaccine was also available for free to students returning to campus in the fall of that year.
Today, undergraduate students are required to receive the quadrivalent meningococcal conjugate ACYW-135 vaccine before matriculation.
In contrast to earlier hesitations against vaccinating students, last fall, the College required students wishing to return to campus to sign a “community expectations” agreement saying that they would become vaccinated against COVID-19 when a Food and Drug Administration and Dartmouth-approved inoculation became available. Vaccination is now required for students starting in the fall of 2021, and outgoing Provost Joseph Helble said in a recent Community Conversations” livestream that the College is finalizing a mandate for faculty and staff as well.
According to Nachlis, the College’s ability to enforce its “community expectations” agreement stems from its authority as a private institution. At public universities, he said, an analogous policy would be influenced by state politics and would vary depending on if a state leans red or blue.
While Wheelock was distrustful of smallpox inoculation in the eighteenth century, Nachlis understands Dartmouth’s community agreement and its requirement that students receive the vaccine as displaying greater trust in science and public health practices.
“I think [the community agreement] shows an admirable and reasonable faith in our public and private regulatory, funding and product development and distribution organs, saying, ‘We expect when the time comes, they’re gonna get it right, and it’ll help our campus to be safe,’ and I think they’ve been proven right,” Nachlis said.
Dartmouth’s vaccine contributions
Vaccines have come a long way since the smallpox inoculations of the late 18th century: The World Health Organization now lists 29 diseases that can now be prevented by vaccines. Dartmouth community members have contributed to a number of these past and present vaccination efforts.
Medicine professor Ford von Reyn’s research has focused on tuberculosis, especially tuberculosis patients with HIV in Africa. Von Reyn’s research led to the 2013 creation of the DAR-901 vaccine, with the “DAR” standing for both Dartmouth and Dar es Salaam, a city in Tanzania where Von Reyn did much of his work on the vaccine. According to the WHO, tuberculosis is the leading cause of death among infectious diseases, killing an estimated 1.5 million people in 2018 alone. Von Reyn said that the only licensed vaccine against tuberculosis is the Bacillus Calmette–Guérin vaccine, which is given at birth.
“[BCG is] the most widely used vaccine in the world,” von Reyn said. “It’s quite effective for 15 or 20 years, but then its efficacy begins to wane, and for that reason, there’s been international efforts of developing a booster — and that’s exactly the role that we see for DAR-901.”
The DAR-901 vaccine has been in the works for decades. According to von Reyn, he started the research in 1984. A previous version of DAR-901, SRO-172, was proven to be effective in a Phase III trial, but the manufacturing method of the vaccine was ultimately not scalable enough to make global production possible, he said. While the newer DAR-901 can, unlike SRO-172, be manufactured at scale, the FDA requires clinical testing of vaccines to return to “square one” if there are changes to the manufacturing process, von Reyn said. A Phase III trial for DAR-901 is “poised to start” in 2022, which, if successful, would support the licensing of the vaccine.
Peter Wright, a professor of pediatrics at the Geisel School of Medicine, has studied existing types of polio vaccines and how they might be improved. According to Wright, polio has been eradicated in all areas of the world except for Afghanistan and Pakistan, where community immunization has been made difficult by war in the region. WHO data shows that there has been a 99% reduction in polio cases since 1988, and there were just 33 cases worldwide in 2018.
This progress can be credited to the success of two polio vaccines: an attenuated vaccine developed by Albert Sabin and an inactivated vaccine developed by Jonas Salk. While both versions have proven effective, the Sabin vaccine can cause vaccine-associated paralytic polio in approximately 1 in 2.7 million recipients of the vaccine — meaning that in extremely rare instances, the vaccine actually gives the recipient the disease. This danger curtails the vaccine’s effectiveness, Wright said, which is why he is working on the development of a new one
“We are embarking...on a trial of a new or novel polio vaccine, derived very closely from the Sabin vaccine but with further attenuation, further genetic stability, and basically designed to overcome some of the limitations that we’re seeing with the Sabin vaccine in the field,” Wright said.
According to Wright, a trial of this new polio vaccine is underway at the University of Vermont and the University of North Carolina, with a third arm of the study to begin at the College this summer.
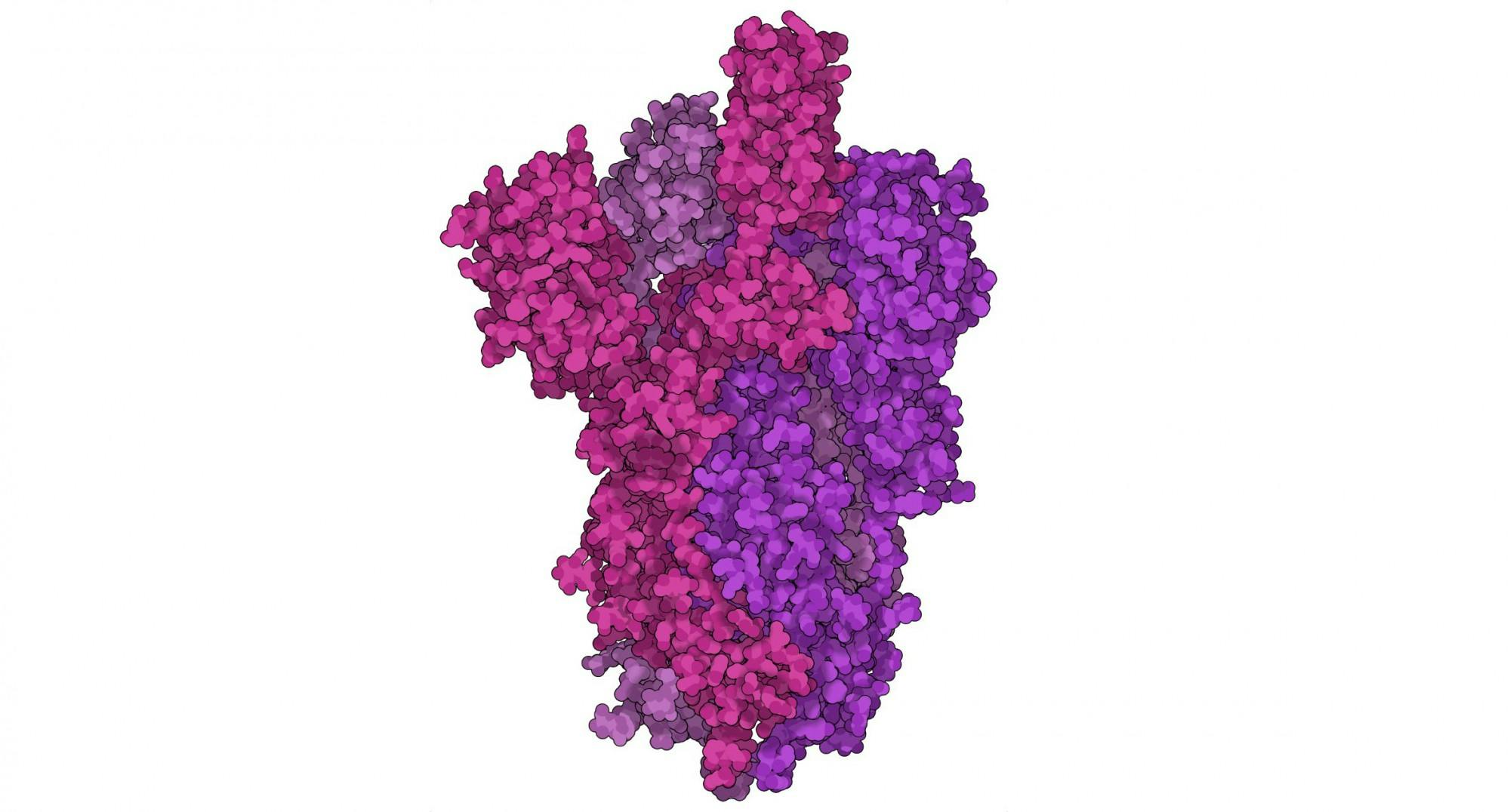
Research initiated at Dartmouth was also instrumental in the creation of COVID-19 vaccines. The McLellan lab, founded at Dartmouth in 2013 by former biochemistry professor Jason McLellan, contributed to developments in coronavirus research that form the “basis for most approved vaccines,” according to postdoctoral fellow in the lab Dr. Daniel Wrapp. Wrapp, who was a doctoral student at the Geisel School of Medicine when he joined the now-Texas-based lab, said that the coronavirus infects new people by invading host cells using a spike protein. After entering a cell, the virus can undergo replication and infect more cells.
“Basically, we want to give the immune system something that looks like spike protein so that if you ever come into contact with the coronavirus, you already have antibodies which are capable of binding to [the] spike and neutralizing the virus before it infects you,” Wrapp said.
However, spike proteins are “notoriously difficult to produce” due to how unstable they are, Wrapp said. According to Wrapp, the lab’s creation of the novel coronavirus’ spike protein was based on the work of Dr. Nianshuang Wang, who was a postdoctoral researcher at Dartmouth when he developed the “first structure” of the spike protein for HKU1, a mild coronavirus, in 2016. According to Wang, the process to stabilize the spike protein took him two years.
“That [development] gets us very well prepared for a new coronavirus that can emerge in the future that we can quickly generate a new vaccine for,” Wang said.
According to Wrapp and Wang, the spike protein developed by Wang enabled the lab to design stabilizing mutations for viruses that have more unstable spike proteins, including the coronavirus that causes MERS, a disease that saw a major outbreak in 2012.
When the lab received word in late 2019 that there was a likely novel coronavirus outbreak in the Chinese city of Wuhan, Wrapp said, they immediately began developing “the equivalent mutations into this novel spike protein.”
The team finished work on the spike protein on Jan. 30, 2020. According to Wrapp, the spike protein developed by the McLellan lab has been incorporated into all three FDA-approved vaccines, of which hundreds of millions of doses have been administered to Americans since December.